- Home Page
- Company Profile
-
Our Products
- Cardiac Medicines
- Telmisartan
- Alcarnit Tablet
- Fenacin PC Tablet
- Furosemide Tablet
- Diltiazem Hydrochloride
- Atocen Tablet
- Lisinopril Tablet
- Losartan Potassium
- Metoprolol Tartrate
- Amlodipine Losartan
- Cendop Tablet
- Amlocen 10 Tablet
- Atocen 20 Tablet
- Atocen 40 Tablet
- Amiodarone Hcl
- Enalapril Maleate
- Hydralazine Tablets
- Nifedipine Extended Release
- Prazosin Hydrochloride
- Ramipril Tablet
- Clopidogrel Tablet
- Trimetazidine Hydrochloride Tablet
- Verapamil 40mg
- Amlodipine Besilate
- Amiodarone HCI
- Nifedipine Tablets
- Clopidogrel Tablets
- Acetazolamide Tablets
- Amiloride HCI
- Pain killer Medicines
- Anti-Bacterials and Antibiotics
- Gastrointestinal Medicines
- Beta Lactam Antibiotics Medicines
- High Blood Pressure Medicines
- Nutraceuticals, Vitamin & Mineral Products
- Oral Suspensions
- Nutraceutical Supplement
- Cardiovascular Medicines
- Anti Cancer Drugs
- CNS Tablets
- Amoxicillin Capsules
- Clotrimazole Creams
- Antimalarial Tablets
- Ointments Cream
- Antiseptic Ointments
- Anti Viral Tablets
- Veterinary Injection
- Antihistamine Tablets
- Anti Ulcer Tablets
- Anti-Infective Drugs
- Alopecia Medicines
- Analgesic And Anti-Inflammatory & Anti Pyretic
- Anti HIV
- Respiratory Syrup
- Sodium Valproate Tablets
- Cetirizine Tablets
- Anti Fungal Tablets
- Anti Biotics
- Antituberculosis Tablets
- GIT Medicine
- Pharmaceutical Medicine
- PANTOCEN-DSR Capsules
- NALTREXONE TABLETS IP 50MG
- CLOTRIMAZOLE VAGINAL TABLETS
- ZICAL Vitamin D3 Tablet
- ROXICEN Tablet 150mg
- MYCOCEN-M Tablet
- MIFEPR-ISTONE TABLETS IP 200MG
- CLOZAPINE TABLETS IP 50MG
- CLOMIFENE TABLETS
- CENVERT-8 Tablet
- CENCORATE 200 Tablet
- ACROCEN-2 Capsules
- ACENOCOUMATOL TABLETS IP 1MG
- NAPROXEN TABLETS IP 500MG
- GUMMIES
- VITACARE- ginger and turmeric immune support gummies
- VITACARE-calcium And vitamin D3 healthy bones And muscles gummies
- VITACARE- VITAMIN C
- VITACARE-vitamin B12 And folic acid gummies
- VITACARE- brahmi And shankhpushpi gummies
- VITACARE-multi vitamin health supplements gummies
- VITACARE- vitamin c And echinacea immunity booster gummies
- VITACARE-Aamla and ashwagandha Enhance endurance gummies
- VITACARE-vitamin c and zinc immunity booster gummies
- VITACARE-Glucosamine And chondroitin gummies
- VITACARE- apple cid-er vinegar weight management gummies
- VITACARE- folic acid gummies
- Cardiac Medicines
- More information
- Contact Us

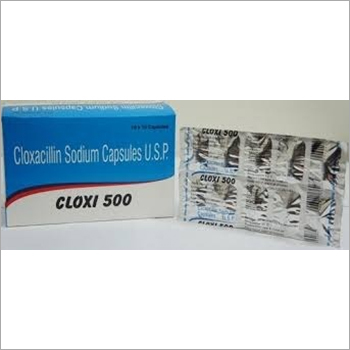
Cloxacillin Sodium Capsule USP
MOQ : 1 Unit
Cloxacillin Sodium Capsule USP Specification
- Salt Composition
- Cloxacillin Sodium
- CAS No
- 61-72-3
- Dosage
- As per doctor
- Medicine Type
- Allopathic
- Molecular Formula
- C19H17ClN3NaO5S
- Pacakaging (Quantity Per Box)
- 10 x 10 capsules
- Grade
- Pharmaceutical Grade
- Origin of Medicine
- Imported
- Usage
- Clinical, Hospital Use, Antibiotic for bacterial infections
- Dosage Form
- Capsule
- Expiration Date
- 24 months from date of manufacture
- Appearance
- Capsule
- Storage
- Dry Place
- Indication
- Bacterial infections responsive to cloxacillin
- Assay
- Not less than 90% and not more than 120% of the labeled amount
Cloxacillin Sodium Capsule USP Trade Information
- Minimum Order Quantity
- 1 Unit
- Delivery Time
- 10 Days
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Amoxicillin Capsules Category
Paracetamol and Caffeine Capsules
Minimum Order Quantity : 1 Unit
Appearance : Capsules
Assay : Paracetamol 500mg, Caffeine 30mg per capsule
Storage : Dry Place
Indication : Headache, migraine, fever, mild to moderate pain
Dosage : As per doctor
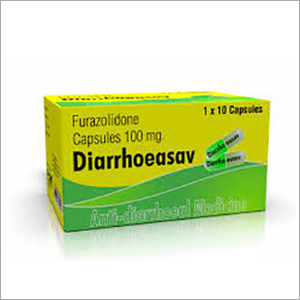
Furazolidone Capsules
Minimum Order Quantity : 1 Unit
Appearance : Yellow Powder (filled in capsules)
Assay : 98% Min
Storage : Dry Place
Indication : Bacterial Diarrhea, Giardiasis, Cholera, Enteritis
Dosage : As per doctor
Fluconazole Capsules
Minimum Order Quantity : 1 Unit
Appearance : Cpasules
Assay : Not less than 98%
Storage : Dry Place
Indication : Fungal infections
Dosage : As per doctor
"We are accepting bulk enquiries and for international enquiries also."

 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese Send Inquiry
Send Inquiry